 Back in April I wrote a blog for the Huffington Post about Lies, Damned Lies and Drug Trials.
Back in April I wrote a blog for the Huffington Post about Lies, Damned Lies and Drug Trials.If anyone doubted what my point was, please read today's story in the New York Times called "Why the Data Diverge on the Dangers of Vioxx."
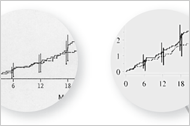
It turns out that the sacred "first 18 months" during which time Vioxx supposedly didn't cause heart attacks (according to Merck) wasn't so sacred.
"The new data Merck submitted to the Food and Drug Administration earlier this month include what happened to the patients for a longer period after they stopped taking the drug. And in this presentation, the Kaplan-Meier chart — the one critics have seized upon — shows those lines starting to separate at around four months and remaining separate thereafter, although they do diverge more widely at the 18-month point."
NY Times continues:
"In The New England Journal of Medicine's response to the original manuscript, the editor, Dr. Curfman, asked the authors to remove the assertion "that increased risk was observed only after 18 months." Nevertheless, the 18-month finding was published as part of the article's main results. But Merck and the researchers continue to insist that the 18-month finding was a legitimate conclusion, based on the data that were analyzed. "
So here we go again. Things are worse than the company reported. Now we know you get a heart attack from Vioxx already after four months use. But let's not beat up on Merck. Perhaps they were just unlucky, and their scientist too sloppy to find this information right away. Or, maybe they had a hunch and tried to hide it. Who knows?
What we do know is that very rarely do companies hide good information and it is much more common for them to behave like five-year olds who just raided the cookie jar when they have bad news to tell. But even when they really try, big companies rarely manage to pull off the lies with the aplomb of a fiver-year old.
So what does the lead author have to say? He responds like a true five-year old:
"If The New England Journal didn't think this was an appropriate and satisfactory product, they didn't have to publish it," Dr. Bresalier said.
No comments:
Post a Comment